Introduction
|
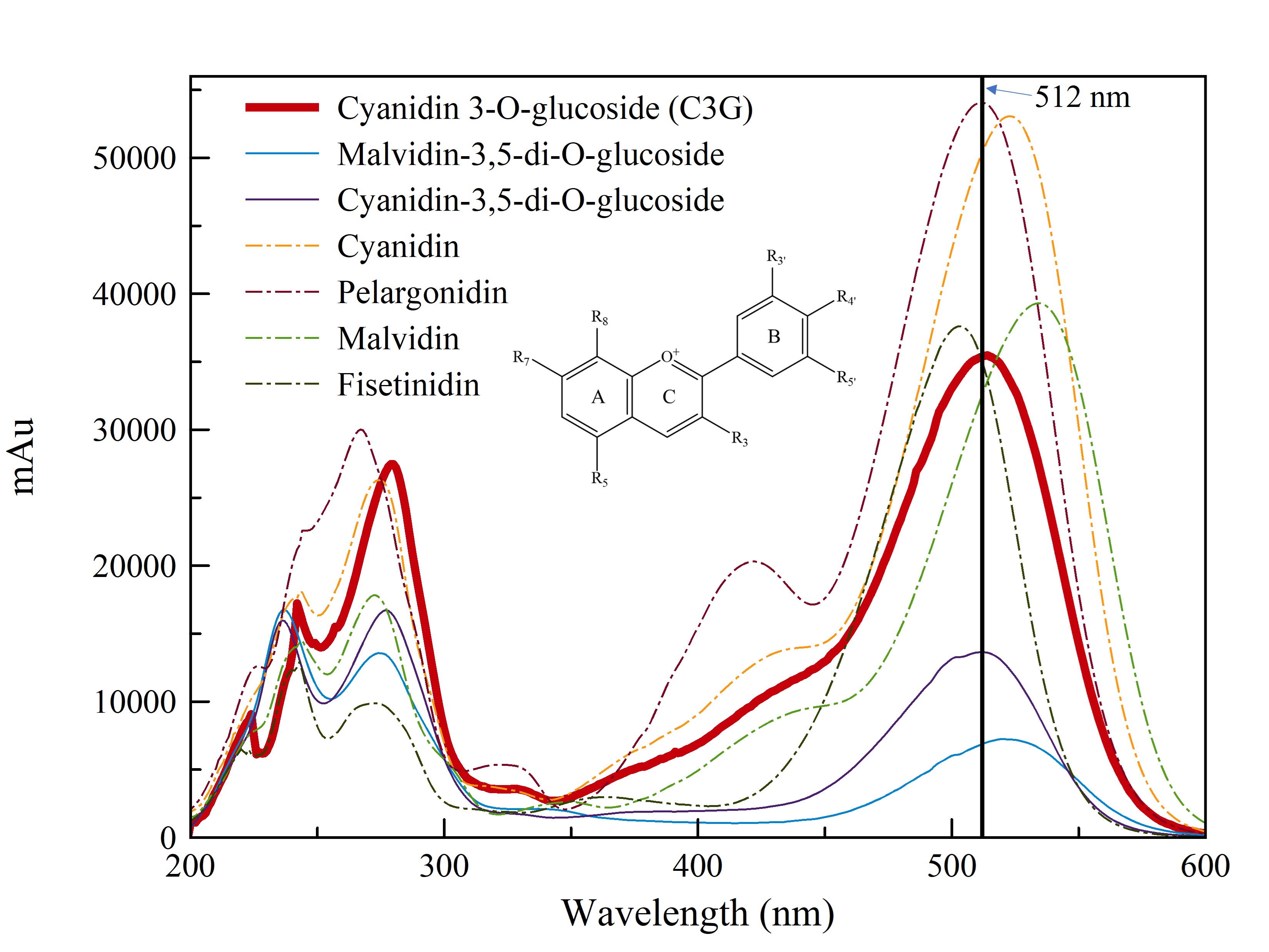
Molar relative response factors (MRRFs) can be used to correct these effects, allowing the inexpensive and accurate quantitative analysis of many anthocyanins with one master reference standard (i.e., Cyanidin 3-glucoside, or C3G) The general rules for MRRF and λmax shift due to the structural variations for anthocyanins is shown below. 
With the MRRF strategy, individual anthocyanin concentration in food materials can be quantified with improved accuracy. (C3G: cyanidin-3-glucoside) 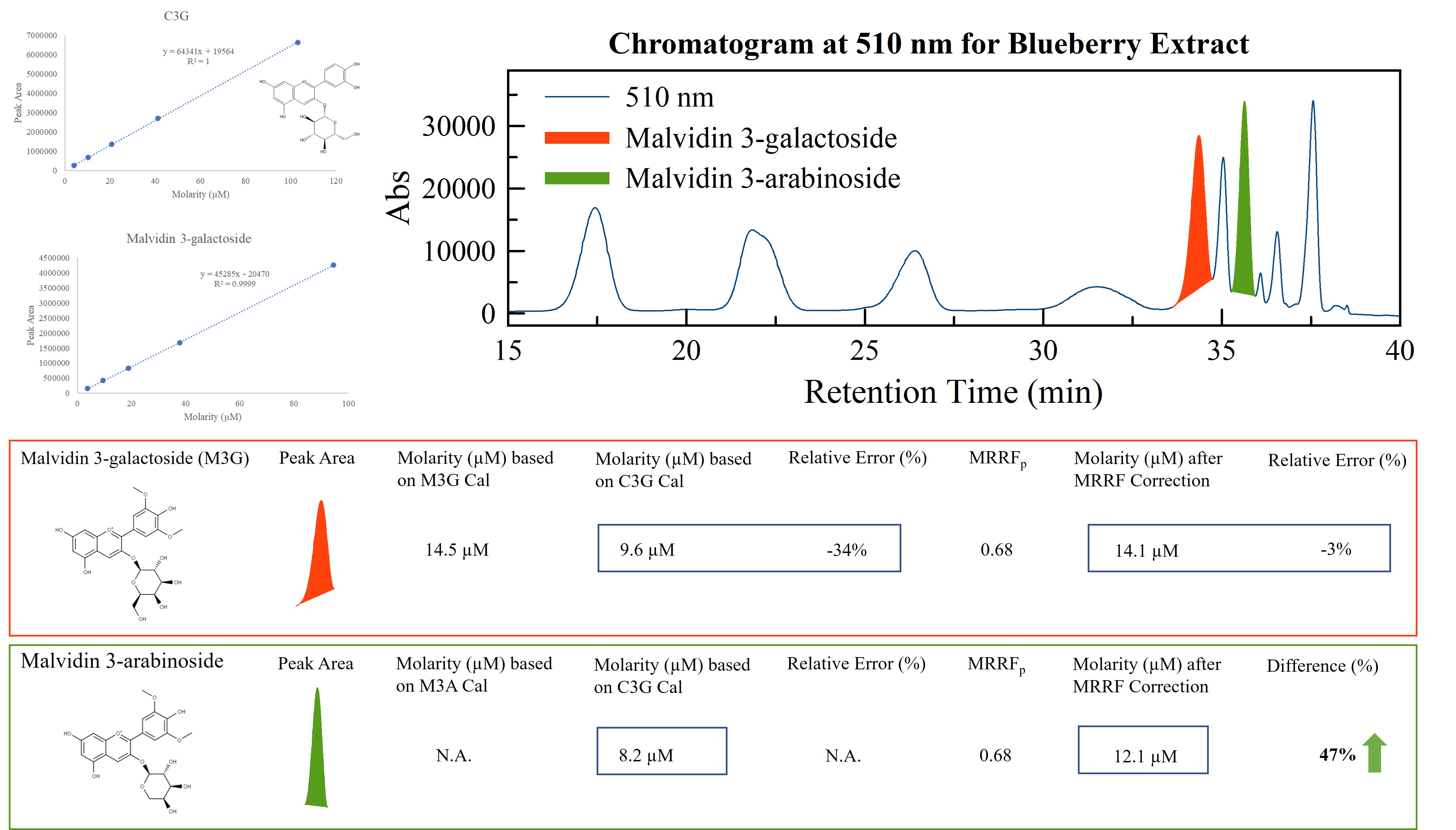
Please search the anthocyanin/anthocyanidin by name, formula search, CAS number, or combined and obtain an output table for matched anthocyanin records with formula, CAS number, MRRF, and links to detailed chemical information for each anthocyanin. |
Database Search
Hint: Please enter keywords in the search box. Compunds with matching names will appear in the list below.Multiple keywords: To select multiple keywords, repeat the process in the Hint. The search function returns the list of plants containing all of the compounds/formulas entered.